Egg based vaccine production12/28/2023 
Alarmingly, the US Centers for Disease Control and Prevention (CDC) reported an increase in influenza A(H2N2)-a strain associated with higher hospitalization and deaths-in recent outbreaks in the USA, highlighting that influenza season preparedness should not be deprioritized ( 5). Concurrently, the COVID-19 pandemic may have affected the circulation of influenza virus in the community, potentially resulting to populations with decreased natural immunity against influenza viral strains or a decrease in viral antigenic drift ( 4). 
As the 2021 to 2022 influenza seasons co-occurred with the pandemic, flu and COVID-19 co-infections have been reported and may have increased hospitalizations with significant mortality as reported in the United States of America (USA) ( 3). The emergence of COVID-19 pandemic in the early 2020 resulted in a decline in the overall reporting and positivity of influenza infection ( 2). Therefore, influenza vaccination programs require constant monitoring and characterization of circulating viral strains for annual formulation. 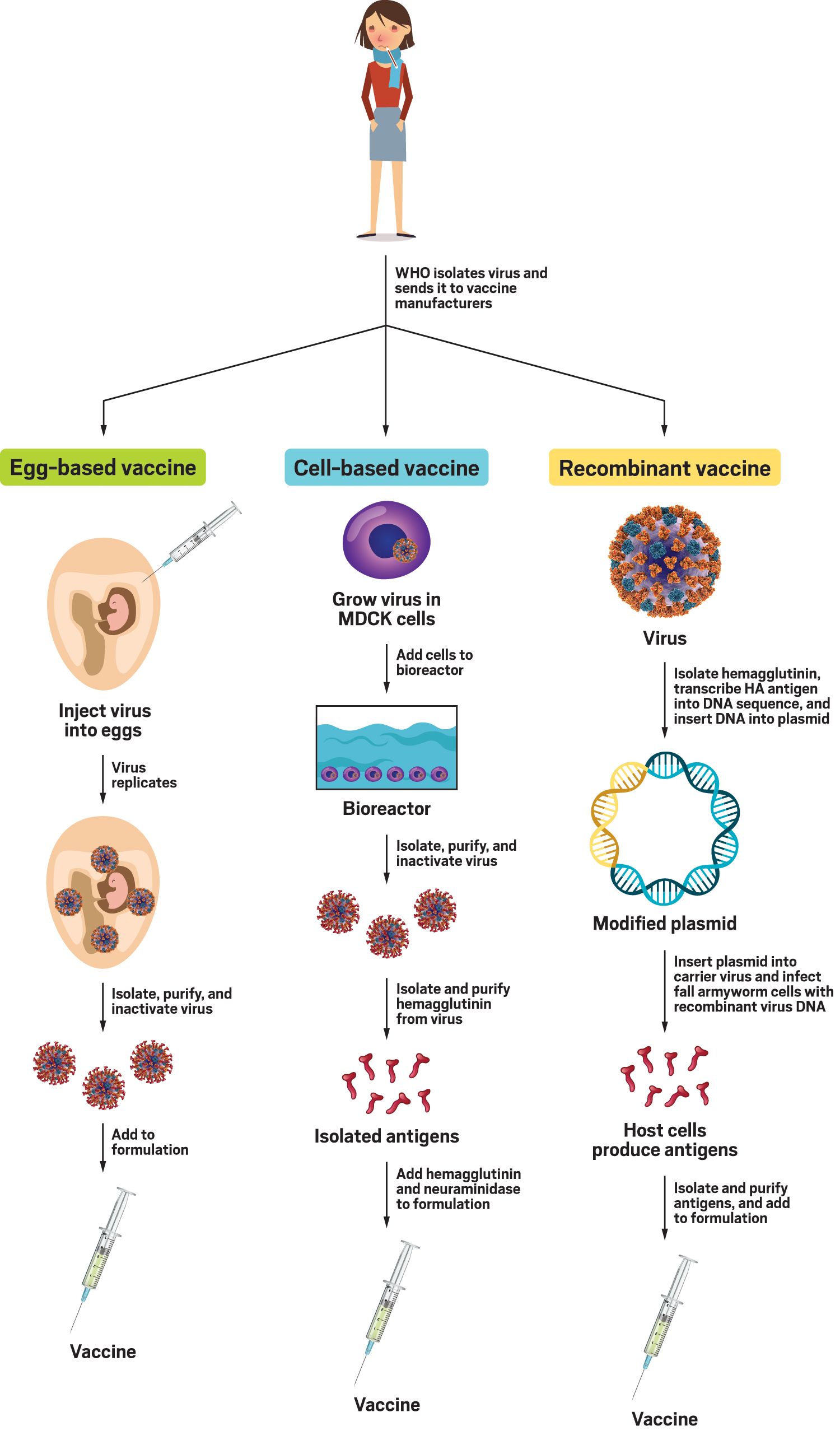
However, conventional vaccination may induce suboptimal immunogenicity and narrow the breadth of protection against the highly variable seasonal influenza strains. The World Health Organization (WHO) indicated that vaccination is the most effective prevention strategy against the infection ( 1). Influenza viruses have extensive genetic and antigenic diversity affecting pathogenicity, host range, and immune evasion, ultimately causing seasonal epidemics and pandemics worldwide. This review also provided discussion points and future perspectives in looking at RIVs as potential universal vaccine candidates for influenza.įlu is an acute respiratory infection caused by influenza viruses. In particular, this review discussed existing knowledge on influenza and vaccine development, current hemagglutinin-based RIVs in the market and in the pipeline, other potential vaccine targets for RIVs (neuraminidase, matrix 1 and 2, nucleoprotein, polymerase acidic, and basic 1 and 2 antigens), and deantigenization process. This review highlights the current progress and advances in the development of RIVs in the context of heterosubtypic immunity induction toward universal vaccine production. With the increasing number of studies and mounting evidence on the safety and efficacy of recombinant influenza vaccines (RIVs), they have been proposed as promising platforms for the development of universal vaccines. An important property of universal vaccines is their ability to induce heterosubtypic immunity, i.e., a wide immune response coverage toward different influenza subtypes. However, given the high plasticity of the virus and the suboptimal immunogenicity of existing influenza vaccines, scientists are moving toward the development of universal vaccines. Vaccination is considered the most effective protective strategy against the infection. Flu, a viral infection caused by the influenza virus, is still a global public health concern with potential to cause seasonal epidemics and pandemics.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
